  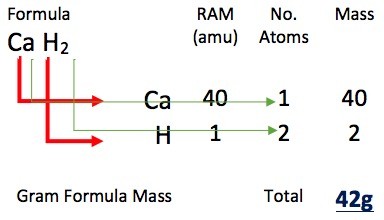
It is soluble in water and decomposes at high temperatures evolving hydrogen gas. Sodium tetrahydroborate, NaBH 4, is a white crystalline compound (decomposes at 400 ☌) usually called sodium borohydride. It is used as a reducing and hydrogenating agent and for dehydrating organic solvents. The hydride dissolves in ethers, and reacts violently with water. Lithium tetrahydridoaluminate, LiAlH 4, is a colorless crystalline compound (decomposes above 125 ☌) usually called lithium aluminum hydride. This hydride is used as a hydrogen gas generator, or a dehydrating agent for organic solvents. Since it dissolves in ethers slightly, the hydride is used as a reducing agent in organic chemistry.Ĭalcium hydride, CaH 2, is a colorless crystalline compound (mp 816 ☌), and reacts mildly with water evolving hydrogen gas. Water reacts vigorously with lithium hydride evolving hydrogen gas. Quantitative evolution of hydrogen gas at the anode during the electrolysis of the fused salt suggests the existence of H. Li + and H - form a lattice with a rock salt type structure. Lithium hydride, LiH, is a colorless crystalline compound (mp (melting point) 680 ☌). Gas chromatography and rotational lines in the electronic band spectrum of H 2 can distinguish two hydrogen isomers. The theoretical ratio of para-hydrogen is 100% at 0 K, but it decreases to about 25% at room temperature, since the ratio of ortho-hydrogen increases at higher temperatures. Since para-hydrogen is in a lower energy state, it is the stabler form at low temperatures. Spins of ortho-hydrogen are parallel and the sum is 1 resulting in a triplet state. Spins of para-hydrogen are anti-parallel and the sum is 0 leading to a singlet state. 
Especially in the case of a hydrogen molecule, the difference of properties is significant. There are nuclear-spin isomers in diatomic molecules of the nuclides whose spin is not zero. \) Properties of isotopic hydrogen and water Properties 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
